The fetus is showing researchers a thing or two about how to fly under the immune system radar.
It was University of Minnesota researchers who first found in 1989 that this burgeoning offspring of a mother and father’s DNA expresses a molecule called HLA-G that could locally silence the mother’s immune response against daddy’s foreign contribution.
Nine years later, Medical College of Georgia research partners pediatric oncologist Dr. David Munn (’84) and immunologist Dr. Andrew Mellor showed that the fetus also expresses the enzyme indoleamine 2,3-dioxygenase, or IDO, to enable it to survive and typically thrive in what should be a hostile environment.
Flash forward to 2016, and the Atlanta-based Carlos and Marguerite Mason Trust, which funded some of the first steps into the potential of IDO and HLA-G to likewise protect a transplanted organ, has just provided $1 million to help a team of MCG physician-scientists and basic scientists take many, many more.
“We want to harness these natural mechanisms for survival and give patients, like the fetus, the optimal opportunity to thrive,” says Dr. Anatolij Horuzsko, MCG immunologist and a principal investigator on the extensive new studies.

“We have had much better results in decreasing acute rejection even with the immune suppressive medications that we have had,” says Dr. Laura L. Mulloy, chief of the Section of Nephrology, Hypertension and Transplantation Medicine who has been at the forefront of transplant medicine for nearly 20 years. “But we have made no changes in long-term graft survival. So there is something going on that we are missing.”
Good deeds by an errant child
Mulloy and Horuzsko are longtime colleagues in their work to fill in some of these important blanks and have early human evidence that HLA-G is much more than a placeholder. Horuzsko calls HLA-G, which clearly downregulates immune activity, an errant child in the human leukocyte antigen, or HLA, family. HLA is a large group of genes that express proteins that sit on the surface of our cells, which help define us and what we have been exposed to in life. This complex of proteins helps our immune sytem differentiate between which cells are ours and which belong to invaders, such as viruses, bacteria and cancer. In fact, HLA-G may be a sort of fail-safe mechanism to help keep the immune system from becoming overzealous in its watchguard role.
While only identical twins raised in the same environment are likely to have an identical HLA match, physicians try to find the best possible HLA match between a donor and recipient. Horuzsko and Mulloy are among the first to measure levels of HLA-G.
They curiously examined a group of patients – 50 with no signs of rejection and 17 with chronic rejection, the biggest cause of transplant failure. They found a clear diference between the two groups: patients who did well had naturally high levels of HLA-G. In fact, they had the most powerful known version of HLA-G, the dimer, where two molecules are actually packaged together. The MCG investigators published the stunning results in April 2014 in the Journal of Immunology Research, and the clear correlation has held up as the number of patients has risen to nearly 200.
“This molecule participates in a whole orchestra called immune tolerance,” Horuzsko says.
“We don’t really know if most of us are expressing HLA-G.” But in transplant patients who do, high levels of this natural downregulator mean patients can do well with a lot fewer prescription drugs designed to keep their immune systems at bay. That’s a good thing, since these drugs carry a whole host of side effects, from increased infections and cancer to actual failure of the transplanted kidney, the immunologist says.

One of the many things Horuzsko and Mulloy are now pursuing is what genes upregulate HLA-G expression and its requisite receptor. They have already identified patients missing the receiving end who clearly did not benefit from higher HLA-G levels. On the other hand, transgenic mice with the human HLA-G receptor and a skin transplant that also are given HLA-G get the same stunning results as the transplant patients the researchers studied.
No doubt something besides a fetus makes HLA-G, but it may be years before they know what that is, Horuzsko says. In the meantime, with the everpresent goal of one day giving HLA-G to patients who aren’t fortunate enough to express more of their own, one of the many things the Mason Trust grant is enabling is the researchers’ continued work in the also likely lengthy effort to identify the most powerful version of HLA-G. They also want to better understand just how it puts a localized kibosh on the immune system.
A newer goal is developing more sophisticated screening to augment decades-old HLA typing with a humanized mouse model.
These mice are missing an immune system, and through a blood transfusion or stem cell injection, they will temporarily assume the immune system of the human transplant patient. Then, by adding the blood of a potential living donor to the mix, the researchers will gain additional insight into how well the two systems can coexist and, consequently, the possibility of long-term transplant success.
“By putting these two immune systems together in a living mouse, we can see beforehand how they interact,” Horuzsko says. He and co-investigator Mulloy also will try different immunosuppressive cocktails to see which works best for the patient the mouse represents.
“It is a surrogate, a way to look at your immune response without directly affecting you,” adds Mulloy. “You are talking about personalized medicine.” While the living-related donor surgery and transplant are scheduled, enabling time for such detailed pretesting, the relatively short notice that a deceased donor organ is available will, at least for the time being, preclude a mouse surrogate.
What kills kidneys
Diabetes and hypertension – which often go hand-in-hand — are the main reasons kidneys die and transplants are needed, says MCG nephrologist Dr. Stan Nahman. “With an aging population adding about 125,000 new dialysis patients to the pool every year in the United States, it’s a big deal,” he says.
The United Network for Organ Sharing manages the kidney transplant and other organ waiting lists. Without a living, related donor, potential transplant patients already wait three to five years in this part of the country. That’s a majority of the patients at AU Medical Center, where 70 percent of kidney transplant patients receive a cadaver donor.
At the Augusta transplant program, most patients are in their early 60s, with fairly equal numbers of men and women, and the vast majority of patients are black. There is a particularly difficult vicious cycle for black patients seeking donors because hypertension and diabetes are particularly virulent in black populations, so many family members may be battling the same disease that caused their loved one to need a transplant. Differences in the percentages of blood types in black and white populations can make identifying a suitable donor – dead or alive – even tougher.
“It’s close to our hearts,” says transplant surgeon Dr. Todd Merchen. “It’s a horrible thing to have people come forward and say I want to donate to my grandmother or to my brother and not be able to.”
Some innovative efforts nationally are taking a serious stab at improving transplant odds. The Food and Drug Administration has on the fast-track a University of California, San Francisco, project that is using nano-engineering to create an implantable artificial kidney designed to mimic both the metabolic and water-balancing jobs of the kidney. It’s slated for clinical trials next year.
Also, paired donor matching works somewhat like crowdfunding, to make more resources, in this case, living kidneys, available in the U.S. It essentially works this way: If someone wants to donate to a family member, but cannot, because of blood type or other incompatability issues, there are national organizations with sophisticated software that can match up these like-minded souls so that an Augustan might donate to a New Yorker who donates to someone in Alabama and so on, Merchen says. “I would donate in a minute to a loved one,” says Nahman.
Glowing in IDO
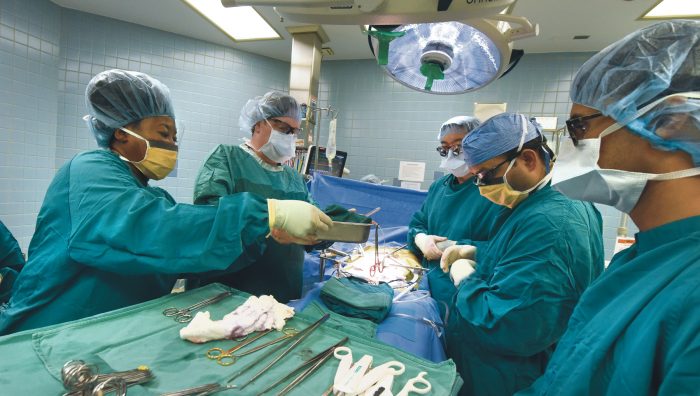
It’s not so much the intricate transplant surgery, first successfully performed by Nobel Prize winner Dr. Joseph E. Murray at Boston’s Brigham and Women’s Hospital in 1954, that concerns Dr. Todd Merchen most, but the response of his patient’s immune system to it. “Your body was created in a way to look at anything that it does not see as itself as hostile,” he says. “That is why we fight off bacteria and cancer.”
Even diminished by today’s immunosuppresive drug regimen, the residual incessant attack by the immune system is a major contributor to chronic transplant rejection.
That’s why Merchen, chief of the Section of Transplant Surgery, adds to his patient-packed days time working in the lab with nephrologist Dr. Stan Nahman and cell biologist Dr. Zheng Dong, both co-investigators on the new grant.
“We want to extend the amount of time people keep their kidney function. We would love to have it last for 30 years,” says Merchen, Mason Distinguished Chair in Transplant Surgery and Mason Trust principal investigator. Because, just like organ sharing networks that are helping more patients today receive a coveted living donor, better success for each patient translates to better lives for many. It is not uncommon for patients to need a second transplant, and Merchen had just done a third transplant the week before. “The longer we get a transplanted kidney to last, the more people get organs quicker,” the transplant surgeon says. Around this part of the country, the wait may be three to five years.
The team already has evidence that in a rat, activation of IDO in the new kidney quietens the immune response and rejection. IDO innovator Mellor, who recently retired from MCG, found several years ago that just inserting submicroscopic beads – which are actually intended to deliver some gene or drug – caused increased expression of IDO, in his case, in a model of rheumatoid arthritis, an autoimmune disease. Merchen and Nahman found the same held true in their rat transplant model and are continuing similar studies in a mouse transplant model.
But they have also moved to Yorkshire pigs. They have already established a rejection model in the pigs by simply doing transplants and are now trying to avoid inevitable rejection by injecting DNA nanoparticles into the donor pig as well as directly into the donor kidney. In their mouse model, they have seen transplanted kidneys express IDO within 24 hours and, in the pigs, 72 hours after injection, Nahman says. He notes they suspect IDO is on the scene much quicker than the timeframes they have measured. They also are looking systemically for IDO expression, and the other great news is that expression appears local, which should significantly reduce side effects of immune suppression if or when IDO is used in transplant patients, Nahman says. A natural follow up, Merchen says, is to continue to follow the pigs longer to ensure that IDO expression truly translates to less rejection.
While IDO and HLA-G are natural, the reality of keeping kidneys away from blood and oxygen for even the shortest time possible, is anything but. Dong likens it to a stroke in the kidney as the millions of kidney filters come to a screeching halt then are asked to crank back up when blood and oxygen return. While a donor organ can be ischemic for hours as it’s transported across town or the country, some level of ischemic reperfusion injury occurs, even when the donor is in the next operating room. Resulting problems can show up months later, Dong says. This initial damage can mean the kidney never again functions optimally and may result in chronic inflammation that ultimately destroys the kidney. “There is a very good correlation between the storage period and later outcomes,” Dong says. Which is why centers such as MCG’s put an outside limit on transport times. Still, there are problems.
Some cells will die outright; others will linger and could rally or die. To further understand the damage and explore potential therapies, Dong is analyzing tissue from living donor kidneys both before and after Merchen transplants them to look for signs of ischemic reperfusion injury, IDO and/or rejection. Later, they will be looking at the impact of cold storage, which slows cell activity, on patients experiencing rejection, again measuring signs of reperfusion injury and correlating those with time the kidneys spent on ice. Ultimately, they want to better understand how ischemic reperfusion injury correlates with chronic kidney failure and how best to intervene.
“If we can prevent that intrinsic cell death, organs and patients will both live better,” Dong says.
As she helps prepare patients for a transplant today at AU Medical Center, Mulloy ensures they understand the good and potentially problematic. But like her colleagues, she knows that even with current limits, a new kidney most often means a better, longer life. “You have 10 dialysis patients today, one is alive 10 years from now. You take 10 transplant patients today, nine are alive 10 years from now,” she says. “Still, we can do better.”