Cannabidiol Study Targets Children’s Intractable Seizures
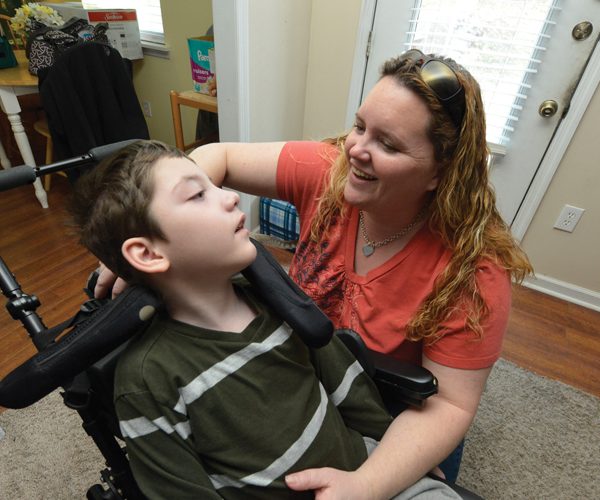
IT WAS just a routine appointment. Valerie Weaver was having her 33-week ultrasound. Her pregnancy had been progressing normally, and she was looking forward to her baby shower the following weekend. She never made it to that baby shower. The ultrasound showed Weaver had reversed diastolic flow, a condition that happens in less than 0.5 percent of pregnancies and causes the reversal of the blood flow through the umbilical artery. “It was like I was taking away from him what my body should have been giving him,” she says.
Preston Weaver was born the next day via cesarean section – a little over 4 pounds of pure perfection.
“He was in and out of the NICU in 20 days,” she says. “We were all shocked.”
Delayed Milestones
SHE TOOK her perfect baby boy home in mid-November, with instructions to follow up regularly with Dr. Jatinder Bhatia, MCG Chief of Neonatology, over the next year. Over the next few months, everything progressed normally . . . until it didn’t.
“I guess it was when he was about 5 months old, I noticed he was missing his milestones,” Weaver says. “I remember him sitting in a Johnny Jumper seat and not having great head or trunk control. He wasn’t grasping at things. He never turned over. Instead of acting like an almost 6-month-old, he was more like a 2-month-old.”
She had expected some delays due to his prematurity. “I honestly didn’t think much of it,” she says. Although he was missing some milestones, to her, he was still perfect – with a little smile that reached his eyes. He’d flash it at her over and over. “I just thought it was the cutest thing.”
But at Preston’s next routine appointment, Bhatia suspected something wasn’t right and referred them to a pediatric neurologist. “We saw Dr. (Elizabeth) Sekul, and she took one look at him and said she suspected those smiles I loved weren’t really smiles at all. They were infantile spasms,” Weaver says. An electroencephalography confirmed the diagnosis and added another – cerebral palsy. “I was honestly more devastated about the CP,” she says. “I had never even heard of infantile spasms.”
Electrical Misfires
ACCORDING to the National Institute of Neurological Disorders and Stroke, infantile spasm is a seizure seen in a type of epilepsy called West syndrome. The spasms, which usually look like a sudden bending forward and stiffening of the limbs or an arching of the back with extended limbs, usually stop by age 5, but are typically replaced by other types of seizures.
The results are catastrophic, often leading to profound developmental regression. That’s because the seizures are a visual sign that something is wrong with the electrical system that controls your brain. “It’s like electrical discharges take over the brain and then the brain can’t control the body,” says Dr. Yong Park, an MCG pediatric neurologist. “And that interrupts cognitive function.”
Essentially, the brain can’t retain information during a seizure. Infantile spasms occur in clusters, and children can have several hundred per day. Treatments are often ineffective.
“Preston was having hundreds of seizures a day,” Weaver says. “We were going to try whatever we could to get his seizures under control.”
Whatever They Could
“WHATEVER they could” has been dozens of medications over his short seven-year life, including steroid injections as an infant that Weaver says made him cry 24 hours a day. Nothing could control the seizures. Doctors even suggested a surgery to separate the hemispheres of Preston’s brain so the seizures couldn’t travel from one side to the other.
“But I wasn’t ready for that,” Weaver says.
They opted, instead, to try the Ketogenic diet, a high-fat, adequate-protein, lowcarbohydrate diet that is often used to treat intractable epilepsies. But by the time they’d decided to try it, the pediatric nutritionist with experience in it had left the Children’s Hospital of Georgia. They would have to wait.
While they did, Weaver,
desperate for something – anything – to help her son, looked for other alternatives. “I was on my computer, researching other drugs morning, noon, and night.”
That’s where she began to read other parents’ accounts of cannabidiol, or CBD, and how effective it was controlling their children’s seizures. “The more I read, the more I learned, the more I believed that this was going to be what helped Preston. I knew this would change our lives.
Cannabidiol is one of over 100 compounds found in the cannabis plant and is nonpsychoactive. Studies in animal models showed that it works as an anticonvulsant, but evidence of the same effect in humans had been mostly anecdotal.

Special Permission
CANNABINOIDS are a family of complex chemicals that lock on to various receptors in the body. Humans have been using cannabis plants for medicinal and recreational reasons for thousands of years, but it wasn’t until the late 1980s that researchers found the first cannabinoid receptor and, later, found that the body produces its own cannabinoid-like chemicals, called endocannabinoids.
Those chemicals bind to one of two cannabinoid receptors, CB1 or CB2. CB1 is mostly found on cells in the nervous system, including certain areas of the brain, while CB2 receptors are mostly found in cells from the immune system.
Scientists believe endocannabinoids and cannabinoid receptors are involved in a vast array of functions, including helping to control brain and nerve activity.
GW Pharmaceuticals, a United Kingdom-based company that has spent a decade sponsoring and publishing CBD research, produces a drug, Epidiolex®, that has been studied at 12 U.S. sites on over 300 children – but only with special permission from the U.S. Food and Drug Administration and Drug Enforcement Administration.
Why?
Because cannabinoid medications are still illegal in most of the United States.
Georgia legislators failed to pass a bill in 2014 legalizing the use of medical cannabis, but lawmakers knew something had to be done. Families of children with untreatable epilepsies were fleeing to other states, such as Colorado, where cannabis is legal.
I HAVE HEARD THE STORIES OF BRAVE GEORGIA FAMILIES DESPERATELY SEEKING TREATMENT FOR THEIR CHILDREN’S DEBILITATING CONDITION. AS GOVERNOR, IT IS MY RESPONSIBILITY TO ADDRESS THE NEEDS OF AND PROTECT OUR STATE’S MOST VULNERABLE CITIZENS, ESPECIALLY WHEN THEY ARE SUFFERING. –GOV. NATHAN DEAL
That something came in the form of a public-private partnership between the state, GW, and GRU after Georgia Gov. Nathan Deal announced his support last April of clinical CBD research and rigorous data collection to “inform and expand the scientific community’s understanding of potential treatments for difficult-to-treat forms of childhood epilepsies.”
“I have heard the stories of brave Georgia families desperately seeking treatment for their children’s debilitating condition,” Deal said at the time. “As Governor, it is my responsibility to address the needs of and protect our state’s most vulnerable citizens, especially when they are suffering.”
Around 466,000 U.S. children have epilepsy and about 20 percent of those have seizures that resist treatment, according to Park. “For these children, it really seems hopeless. There are other options, like alternative diets, but existing medications don’t work.”
The resulting clinical trial – the Epidiolex trial – has been particularly challenging in light of the fact that the medication is derived from marijuana, which is illegal in most of the country.
Above and Beyond
“THIS WAS a very different process [than implementing other clinical trials],” says Dr. Michael Diamond, GRU Vice President for Clinical and Translational Sciences and Senior Vice President for Research. “Normally when you serve as a clinical site for a pharmaceutical trial, there are protocols in place to do things like track progress. We had none of that.”
Challenges included importing the medication, obtaining permission from the DEA and FDA and the Georgia Board of Pharmacy to prescribe it, amending doctors’ prescriptive authority (most can prescribe class II through V drugs; CBD is class I), and tracking children’s progress before and while they are on the medication.
Overcoming the obstacles required no small dose of ingenuity – for instance, the researchers solved the latter problem by commissioning development of a mobile app. But the extra work has been nothing short of a labor of love, Diamond says.
“We wanted to go above and beyond to ensure the integrity of the trial here and for future sites,” Diamond says. “The fact that we were chosen to lead this effort in the state indicates a tremendous amount of trust and confidence by the Governor in our ability to meet this need for Georgia children. This is cutting-edge research, and it’s being done right here in Augusta.”
The final challenge was meeting the study deadline. Officials hoped the trial would be up and running by 2015 in Augusta with planned expansion to Savannah and Atlanta.
The initiative is right on schedule. Preston received his first dose of Epidiolex on Dec. 30 and will be monitored over the next year. Researchers plan to enroll 51 more children.
WHAT I HOPE FOR PRESTON, AND FOR ANY OTHER CHILD WITH INTRACTABLE EPILEPSY, IS THAT HE HAS THE BEST QUALITY OF LIFE HE CAN. –VALERIE WEAVER
Weaver is proud her son, Preston, is making history as the trial’s first participant. “What I hope for Preston, and for any other child with intractable epilepsy, is that he has the best quality of life he can,” she says. “I want him to be able to retain enough to maybe be able to communicate with us in some way, to be able to eat food and taste and enjoy it, and to have a day without pain.”
CBD’S EFFECTS on the BRAIN
GW Pharmaceuticals, the company partnering with GRU in the cannabinoid study, cites several benefits of the drug. Specifically, its decade-long research findings suggest that CBD: n Modulates the flow of ions in and out of cells, especially calcium, which may contribute to its anti-convulsant effects and restrict the intensity and spread of a seizure. n Targets the underlying brain inflammation associated with seizures. n Exhibits neuroprotective qualities, unlike anti-convulsants that are thought to impede cognitive function by reducing useful neural plasticity. “Our hope is that CBD’s early promise of [seizure control and mild side effects] in children with drug-resistant epilepsy may be born out in the formal regulatory studies being undertaken,” says Dr. Geoffrey Guy, Chairman of the GW Pharmaceuticals Board of Directors.