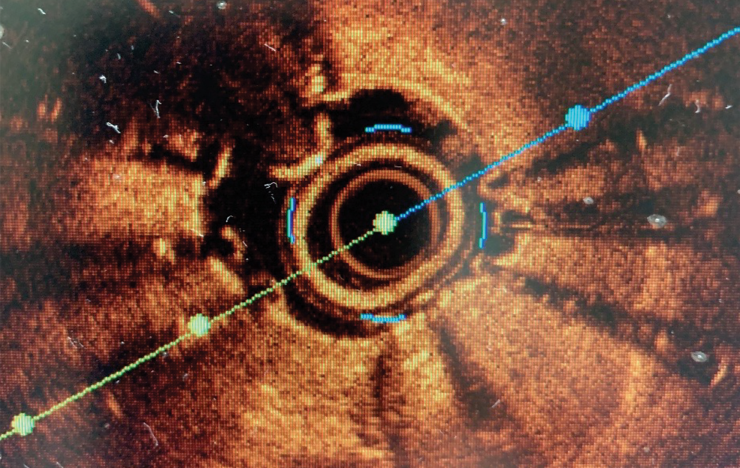
Light waves bouncing around the cavernous confines of the coronary stent placed just months before paint a picture that is oddly beautiful and potentially deadly.
Not unlike those cement pipes placed inside the dirt tunnel running under your driveway to enable water to keep moving without collapse of that essential passageway, this stent was placed to keep this section of the coronary artery open and adequate blood flowing to the heart.
But even the casual observer can tell from this inside view of the artery that the new passageway for blood is now a trickle.
It’s called in-stent restenosis, a life-limiting reality for up to 10% of patients who have stents in their coronary arteries, a frontline approach to treating heart disease, the state and nation’s number one killer.
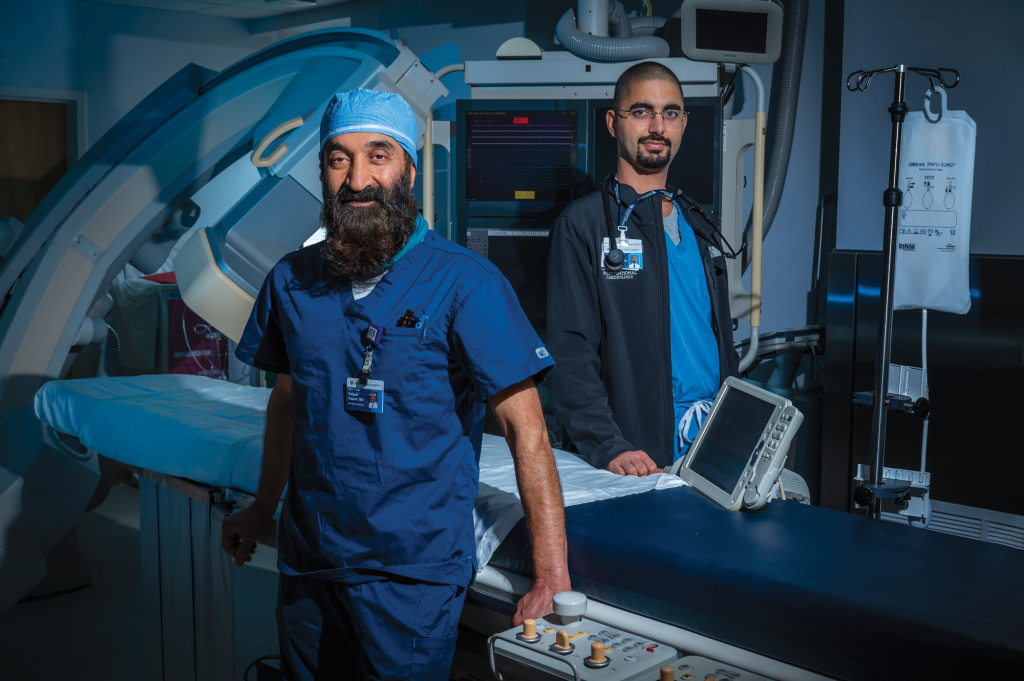
Now Medical College of Georgia interventional cardiologists Drs. Deepak Kapoor and Musa Sharkawi need to make a new path, so they use lasers and diamond-head drills that can blast their way through the scar tissue that formed in response to machinations of the blood vessel needed to place the stent initially. Scar formation can be excessive in some individuals and in short order can have the consistency of hard rubber, and intruders like calcium, cholesterol and fatty deposits can reestablish themselves inside artery walls and worsen the problem. Once these culprits have been pulverized, the doctors use an inflated balloon to create a wider passageway, including expanding the walls of the wire-mesh stent, which was severely under-expanded when originally placed inside the vessel, a common cause of restenosis.
The final step pairs the heart doctors with radiation oncologist Dr. Catherine Ferguson and medical physicist Dr. Ahmad Al-Basheer to deliver a focused dose of radiation directly to their worksite in the form of beta particles, high energy electrons that can, as they do with the wild growth of cancer cells, help stop future overgrowth of scar tissue, which in turn can encourage plaque reformation.
Early Growth
There were just a handful of interventional cardiology training spots in the country in the 1990s when Kapoor was finishing medical school at Lady Haringe Medical College in India and starting his internal medicine residency at Coney Island Hospital in Brooklyn. Physicians in the United States had a bit more than a decade of experience with coronary angioplasty, a technique to reestablish good blood flow to the heart by reopening clogged arteries, as an option for some to the standard of bypass surgery, which was pioneered more than 30 years earlier. Kapoor would do a cardiovascular disease fellowship at the Coney Island Hospital and Cornell Medical Center then move to California to practice cardiology until he got the call from one of those early fellowship programs in New Jersey at St. Michael’s Medical Center, a major teaching hospital for New York Medical College, with an unexpected opening.
Robust prevention strategies, like lipid-lowering drugs, were not commonplace in those early days, but smoking was along with heart attacks. So was having a heart surgery team always on standby during angioplasty, and interventional cardiologists sleeping in the hospital afterward because there was about 50-50 chance that the artery they just opened would close again within a few hours.
Coronary stents, made of a thin, flexible metallic mesh resembling chicken coop wire that was strong enough to shore up those collapsing vessels but malleable enough to adapt to the natural tortuosity of the arteries, surfaced in response.
But there were still problems as these foreign objects prompted excessive scar formation in about one-third of patients and sometimes “turbo charged” the disease process that had originally narrowed the artery.
Coronary brachytherapy centers began to pop up in response to the increasing use of stents and their problems. Radiation is known for its ability to kill growing cells in cancer, and this too was abnormal, undesirable cell growth, and brachytherapy put a sealed radiation source right next to the problem.
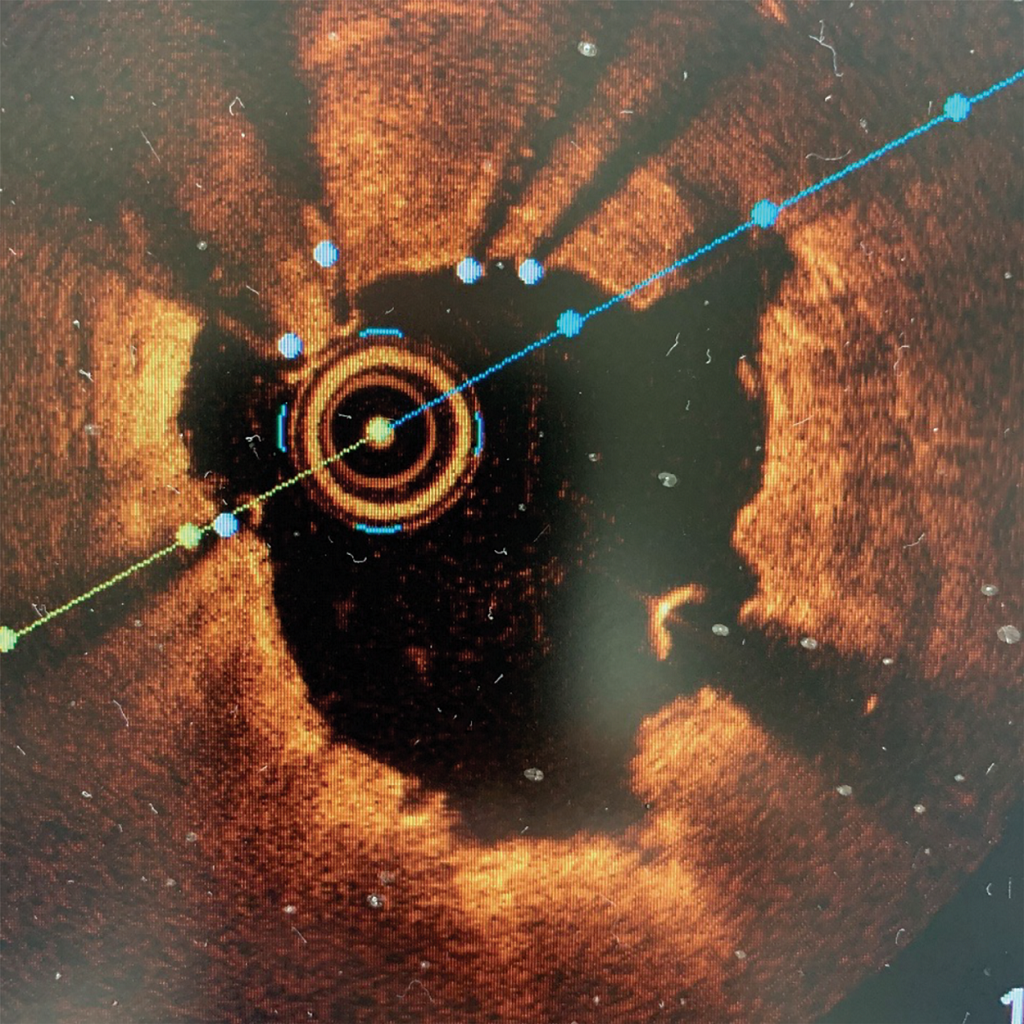
In 2003, the Food and Drug Administration approved drug-eluting stents, designed to help avoid in-stent stenosis. These stents are coated with an antiproliferative drug, the first ones with the chemotherapy drugs sirolimus or paclitaxel, and were shown to reduce major adverse cardiac events, like restenosis and heart attack.
And most patients did fine, as the drug coating to help minimize the overresponse of the healing blood vessel dissipates about the same time the blood vessel is healed. But 5-10% of patients still experienced in-stent restenosis and with the number of stents being used exploding even a small percentage became a big number of patients, Kapoor says.
The rise, fall and rise of coronary brachytherapy
Most coronary brachytherapy centers, including the one at MCG and AU Medical Center, shut down in response to the promise of drug-eluting stents. But there was still that growing number of patients in whom the same symptoms resurfaced, like chest pain, shortness of breath, nausea, weakness, irregular heartbeat, sweating, even a heart attack, within just a few months of their procedures. They found themselves caught in a vicious, sometimes painful and deadly cycle of recurring blockages and repeated procedures to clean out their stents.
Dr. Vincent J.B. Robinson, now co-director of nuclear cardiology and Glen E. Garrison, MD Distinguished Chair in Cardiovascular Medicine at MCG, saw that cycle with his patients. He vividly remembers one patient who had single vessel disease, had repeated bouts of in-stent restenosis and who ultimately was referred for bypass surgery by a colleague while Robinson was out of town. The patient even developed restenosis following his surgery, which is not uncommon, but that was a final straw for Robinson.
At that moment, the closest coronary brachytherapy program was at Louisiana’s Ochsner Medical Center.
Kapoor, director of the cardiac catheterization laboratory, who came to MCG in 2002 was at first noncommittal about restarting a program in Augusta, but Robinson was characteristically committed to the cause and Kapoor is grateful he was. In 2015 the program restarted and today, as one of less than 40 programs nationally, is a regional referral center for these complex, sometimes last-resort cases.
In the many days since, Kapoor and Sharkawi, who joined the team this past summer after completing interventional cardiology and structural heart and peripheral vascular interventions fellowships at Harvard’s Brigham and Women’s Hospital in Boston, have pretty much seen it all.
By the time patients get to the MCG doctors, they’ve typically and rightly had all the more conventional options tried. “People use lasers to melt the plaque then balloon it, for example, and in the cases we get, they’ve already done all these,” Kapoor says.
They sometimes have multiple stents, stacked one inside the other like Russian stacking dolls, in a configuration called a ‘stent-witch,’ in an attempt to keep blood flowing through problematic stents. This stacking may give an initial good result but ultimately and mostly it increases the risk of more blockage, says Kapoor, who has developed expertise in reducing the impact of this ‘stent regret,’ by using lasers to expand the stents, and so the artery’s lumen, and/or a diamond-tipped drill to cut through the now problematic metal layers. In 2019, he published the largest experience with this approach called rotational atherectomy, which is more typically used to break up calcified plaque, to also get rid of these unsalvageable stents. The metal, like the calcified plaque, gets pulverized into minuscule pieces, sludge really, with a particle size of less than 5 microns, small enough to make their way through capillaries or gaps in the cells lining the arteries.
“We are talking about patients who have tried everything and failed,” he says. The coronary brachytherapy team can provide long-term relief to about half those individuals.
At this point Kapoor can pretty much tell who those patients are. Surgery may be an option for those who aren’t, unless excessive stenting has left no good and clear place to bypass and/or the artery has shrunk in response.
That day Kapoor received a referral from a well-known Alabama cardiologist with a patient who had stents placed in the left main artery, which supplies blood to about 70% of the heart muscle. This major blood vessel, typically the heart’s largest, is called the “widow maker” because of the damage it can do when blocked. This patient developed in-stent restenosis and experienced a heart attack despite multiple conventional therapies, which help the vast majority of patients. The referring cardiologist was thinking that perhaps brachytherapy could help, and Kapoor agreed.
Start with a clearer picture
One of the studies the MCG team is pursuing is better understanding the mechanism of restenosis with the newest generation of drug-eluting stents that young Sharkawi trained on. They are finding, perhaps not surprisingly, how essential proper sizing and placement are to ongoing patency. As an example, if a blood vessel has a diameter of four millimeters and you put one in half that size, “you go from a large pipe and it becomes very small then it becomes large again,” Sharkawi says of undersized stents, a site of turbulence, inflammation and risk.
So the team takes extra steps to get the lay of the land. Whether it’s a new stent or opening a blocked one, they get their first look at the blockage during the usual cardiac catheterization procedures. But they also take a second and closer look with intravascular imaging. This includes vascular ultrasound and optical coherence tomography, which instead of soundwaves uses near infrared light, to assess the size of the blood vessel, get better insight on the size and content of the blockage, look for blood clots, which are a major cause of heart attacks, look at the condition of the stent(s) already there and identify the biggest stent that will comfortably fit and the best place to put it once they have cleaned away as much of the obstruction as possible. Sometimes the blockage is so critical that they must first clear a path for the imaging catheter.
Afterward, they look again with angiography, or X-ray, then take one more look with the even more sensitive ultrasound to make sure they did what was needed and that the stent is properly expanded.
These are extra steps, in terms of time (maybe 15 minutes), training and cost, but Kapoor and Sharkawi agree they are essential steps.
The length, circumference and twists and turns of everyone’s arteries are just different, and blockages also come in different thicknesses, lengths and compositions, with some literally as hard as a rock, Sharkawi notes.
“We can’t assume that everybody has a 3 millimeter size right coronary artery,” Sharkawi says. He referenced a course he attended where 200 interventional cardiologists were all asked to guess the size of a coronary artery based on an X-ray. 99% said it was that 3 millimeters — the mean coronary artery size — when it turned out to be 4.5 millimeters, an underestimation that could leave a stent “floating in air” and the patient at high risk for more trouble. That’s why intravascular imaging is considered the standard of care for MCG interventional cardiologists taking care of patients at AU Medical Center and a training standard for future interventionists in the fellowship program at MCG and its teaching affiliate AU Health System, led by Dr. Vishal Arora.
The icing
Once Kapoor and Sharkawi have assessed the problem and cleared a new path, Drs. Ferguson and Al-Basheer, already present in the control room of the cardiac catheterization lab, quickly calculate the radiation dose needed.
The treatment time and dose are individually calculated in real time based on the size of the area that needs treatment — including a 5 millimeter margin — and the rate of decay of their radiation source, says Ferguson.
It’s the nature of things, AlBasheer says, for the energetic electrons they deliver to want to go to a more stable state and they release energy to do that, in a process called decay, which starts the moment the element is born.
To get this internal radiation source precisely where it’s needed, the radiation experts put their catheter inside the interventionalists’ catheter, then water pressure pushes the radiation source directly to the site and keeps it there for the appropriate “dwell time.” After a treatment time of about five minutes, the water stream is reversed to push the radiation source back into a container.
“We treat every part of the artery that we think is susceptible to closing up again,” Sharkawi says, both inside and along either edge if there is any sign of problem there.
Like with tumors, the radiation will help shrink up any vestiges of scar tissue and other impediments left inside the stent and, also as with tumors, deter future regrowth from overhealing.
“We are killing the microscopic, subintimal layers of cells that could over-heal and cause that scar tissue again,” says Ferguson. “Radioactivity when it hits tissue creates free radicals that damage the DNA involved in cell division and that is what kills the cell,’ she says. Kapoor and Al-Basheer both like the analogy of removing weeds then putting down a weed control product to help them stay gone.

The Problem
Placing stents technically injures the blood vessel wall, and normal processes of healing mean platelets move in, inflammation occurs and typically quiescent vascular smooth muscle cells, which normally give a blood vessel strength and flexibility, begin to migrate into and thicken the tunica intima, the innermost layer of the blood vessels, a layer normally comprised of endothelial cells and a thin layer of connective tissue that helps keep blood moving. The vascular smooth muscle cells begin secreting things like growth and pro-inflammatory factors. Bottom line, the lumen, the passageway for blood, shrinks in response.
Stents that are undersized, never fully expanded and/or when layers of stents are added to try to correct the problem, all increase the risk of in-stent stenosis.
So does not properly addressing the original blockage, like those rock-hard calcium deposits, Kapoor says, using the analogy of putting up a building without digging a proper foundation. One result can be an hour-glass shaped stent which clearly does not allow optimal blood flow and gives plaque another good foothold, and/or a diseased segment left just beyond the stent can become a point of stagnation, Kapoor says.
The interventional cardiologists note that even when everything is done right, things can still go wrong. Some people simply scar more in response to the maneuvers needed to clean out the original artery and position the stent. This time Kapoor analogizes the wildly divergent responses by two individuals to the COVID-19 virus. “All of our bodies are different,” he says.
But bodies are always about balance. The endothelial cells that line the miles of blood vessels in our body also fairly quickly embrace the stent, growing through and to the mesh walls. “You want it to be a snug fit,” Sharkawi says.
Radiation Redelivered

Heart disease is the number one killer in Georgia and the nation and heart surgery, angioplasty and atherectomy, typically all make the cut for some of the most common procedures.
At the most recent count there were 36 heart, vascular and radiation centers in the nation and Florida’s first was under development in Winter Haven.
At AU Medical Center, radiation is applied through the Beta-Cath™ System.
This type of internal radiation, or brachytherapy, has Ferguson, Al-Basheer and occupational health and safety technician David Miller patiently on standby in the catheterization lab with Kapoor and Sharkawi. Maggie Corley, cardiovascular nurse clinician, works behind the scenes to coordinate the process.
Radiation is all around us, Al-Basheer reminds us, in the cosmos and the ground, and the radiation they are delivering is very controllable.
Interventional Timelines
 The first coronary angioplasty was performed on an awake patient by German Radiologist Dr. Andreas Gruntzig in 1977 in Zurich, a decade after the technique was used in a leg artery. And it was performed for the first time a year later in the United States by Dr. Simon H. Stertzer at Lenox Hill Hospital in New York. Dr. Jacques Puel implanted the first stent in a human coronary artery in 1986 in Toulouse, France. The Food and Drug Administration approved stents in the US in 1994. The first drug-eluting stent was implanted in a human coronary artery by Dr. Eduardo Sousa in 1999.
The first coronary angioplasty was performed on an awake patient by German Radiologist Dr. Andreas Gruntzig in 1977 in Zurich, a decade after the technique was used in a leg artery. And it was performed for the first time a year later in the United States by Dr. Simon H. Stertzer at Lenox Hill Hospital in New York. Dr. Jacques Puel implanted the first stent in a human coronary artery in 1986 in Toulouse, France. The Food and Drug Administration approved stents in the US in 1994. The first drug-eluting stent was implanted in a human coronary artery by Dr. Eduardo Sousa in 1999.
Today’s stents have thinner struts providing structural support, which means less metal in patients and a reduction in restenosis, and they are the primary method for intervention in patients with one to even three diseased coronary arteries.
A study published in 2020 in the Journal of the American College of Cardiology that looked at a national cohort of more than 5 million patients indicated that about 1 in 10 interventions in the US are for instent restenosis and about 1 in 4 of those patients presented with an acute myocardial infarction.
Stents were old news by the time 31-year-old Sharkawi even thought about interventional cardiology. He grew up in eclectic Dubai, and went to medical school at the Royal College of Surgeons in Ireland, where he spent time in small community hospitals and worked alongside a general practitioner caring for patients living among the iconic, towering Cliffs of Moher. It was the opportunity to see patients at both ends of the spectrum, like those with recurrent in-stent stenosis in need of complex and immediate intervention, and others with long-term management of their heart disease, that drew him to cardiology and to interventional cardiology.